Sponsored Content

By John Rumpakis, OD, MBA
June 30, 2021
I have been speaking and writing about economic analyses of ocular disease states for 35 years. Success in practice is based on having a proprietary skill set, a fundamental population need or demand, appropriate clinical tools and an enlightened and educated clinical team. The diagnosis and treatment of ocular surface disease (OSD) has been the focus of my analysis and articles many times over.
Why, you ask? Because the diagnosis and treatment of OSD requires our proprietary skill-set, clinical acumen and it has one of the greatest rates of prevalence in the population.
Yet I get challenged by colleagues about my financial modeling because they are not seeing those results in their practices. Being challenged is a good thing; it reveals assumptions being made in a model and allows for better understanding of how to communicate touch-points for implementation success.
An assumption I made in my models is that someone on the practice clinical team, whether doctor or staff, was having a pointed discussion or using a qualified questionnaire to uncover signs and symptoms of OSD, rather than just asking, “How are your eyes doing?” The problem with this assumption is that we don’t ask any questions at all because the patient doesn’t mention it, and the patient doesn’t proactively ask because we are not asking the right questions; the proverbial chicken and egg dilemma. Fortunately, this is an easy process to change.
To capture patients who are suffering from chronic disorders such as OSD, we must do a better job in widening the top of the patient capture funnel. The key to that is to ask every patient on every encounter specific questions about the signs and symptoms of OSD. Every patient should be provided with a qualified questionnaire about dry eye. Pick your favorite, whether it is the Standard Patient Evaluation of Eye Dryness (SPEED), OSDI, DEQ5, or any of the others that are out there. Make this a mandatory part of your practice paperwork that a patient is required to complete.
If a patient has a positive sign or symptom related to the questionnaire, have a standing order for point-of-care testing to be done for both inflammation and osmolarity, optometry’s new vital signs as they are called. In doing this one simple step, you have effectively widened the top of the funnel and captured those patients who are symptomatic of OSD, and by performing the point-of-care testing as a standing order, you are now fulfilling one of the components of medical necessity for further dry eye evaluation.
Testing for inflammatory markers and osmolarity have been around for years, and while these tests in and of themselves are not big profit centers in your practice, they are key drivers in proper patient identification and can drive the entire diagnostic and treatment paradigm.
It has been nearly six years since the InflammaDry® test was approved for detecting elevated levels of matrix metalloproteinase 9, a clinically relevant inflammatory marker, in the tears of patients with dry eye disease. If you don’t know, InflammaDry is a single-use test that requires no additional equipment to administer or interpret results. It takes less than two minutes to complete and results are available in 10 minutes, allowing a treatment plan to be established with the patient during his initial office visit.
The CPT code for InflammaDry is 83516 and is defined as follows: “immunoassay for analyte other than infectious agent antibody or infectious agent antigen; qualitative or semiquantitative, multiple step method.”
It is a CLIA-waived test that can be performed in optometrists’ offices where the practice has a clinical lab designation and a physician with the practice has been registered as a clinical lab director. It is a tech-administered test, and the current national payment amount for this lab test is $11.53 per eye. CLIA-waived point-of-care tests are generally not subject to co-pays or deductibles, and should be billed with the QW modifier. Some commercial carriers may not require the QW modifier.
One of the most important aspects about these tests is the ability to have a standing order for them to be performed if the patient presents with a sign or symptom from a qualified questionnaire. And if you are following my recommendation about having every patient, on every visit fill out the questionnaire, you will find that you have many more dry eye patients in your practice than you thought – your funnel just got wider…
Let’s examine now the economics of widening the funnel to your OSD practice.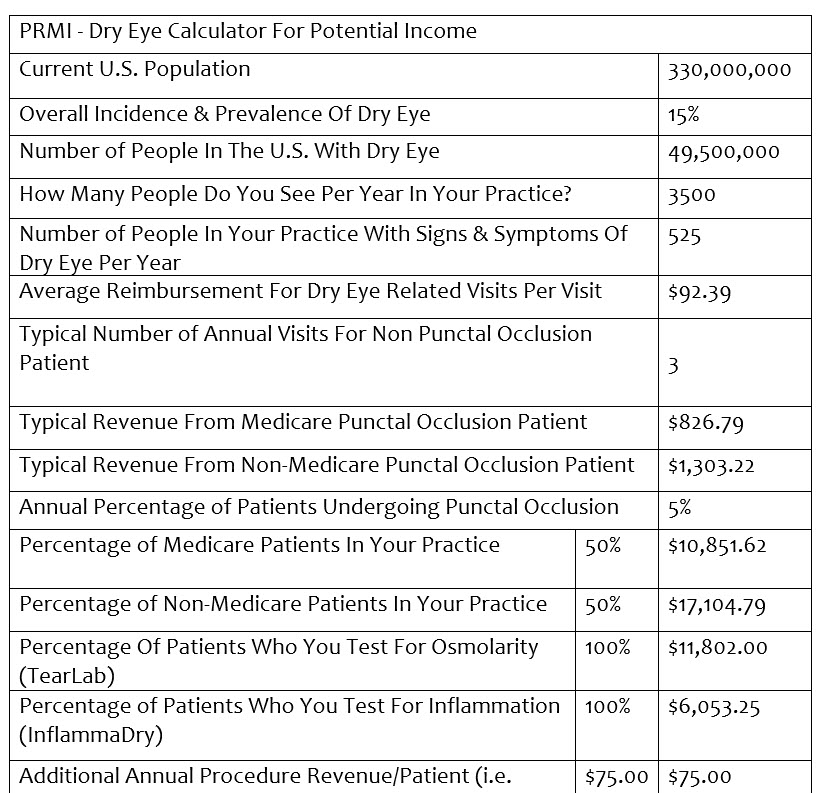


Please note, I didn’t calculate any photography, meibography, lid debridement, intense pulsed light (IPL), neutraceutical recommendations or long-term (multi-year) income, etc., into my calculations because I wanted to be extremely conservative in my approach. I also used national average values for procedures based on the 2021 CMS fee schedule, knowing that while they are significantly below market rate, they are public data for reference purposes.
So why is $298,425 a significant figure? The average annual income for an optometrist in the U.S. today is well below $200,000, and the median income is less than the average. If you can add a single sub-specialty, that has built-in demand, known incidence and prevalence, doesn’t require a heavy front-end investment to incorporate, and deliver high levels of patient satisfaction, reduce your optical redo rate, and can provide a 100 percent increase in income, then it becomes attractive to consider incorporating into your practice.
Other Articles to Explore
Widen That Funnel
Only you can facilitate change in your practice. If you want to see a meaningful change in the number of OSD patients you diagnose and treat, you have to make the changes necessary to find to those patients and get them under your professional care. Then and only then will my financial models be relevant and pertinent to your practice. Widening the top of the funnel and eliminating the “chicken and egg” problem means that you are doing your best in properly identifying your patients, getting vital clinical data, and having the ability to bring them the best in clinical care to help manage this potentially debilitating disease and improve their quality of life.
 John Rumpakis, OD, MBA, is Founder & Chief “Make It Happen” Officer of Practice Resource Management, Inc. To contact him: John@PRMI.com
John Rumpakis, OD, MBA, is Founder & Chief “Make It Happen” Officer of Practice Resource Management, Inc. To contact him: John@PRMI.com
*Article sponsored by Quidel Corporation. Dr Rumpakis was paid for his participation.